Our Technology
Alginate Transmucosal Film
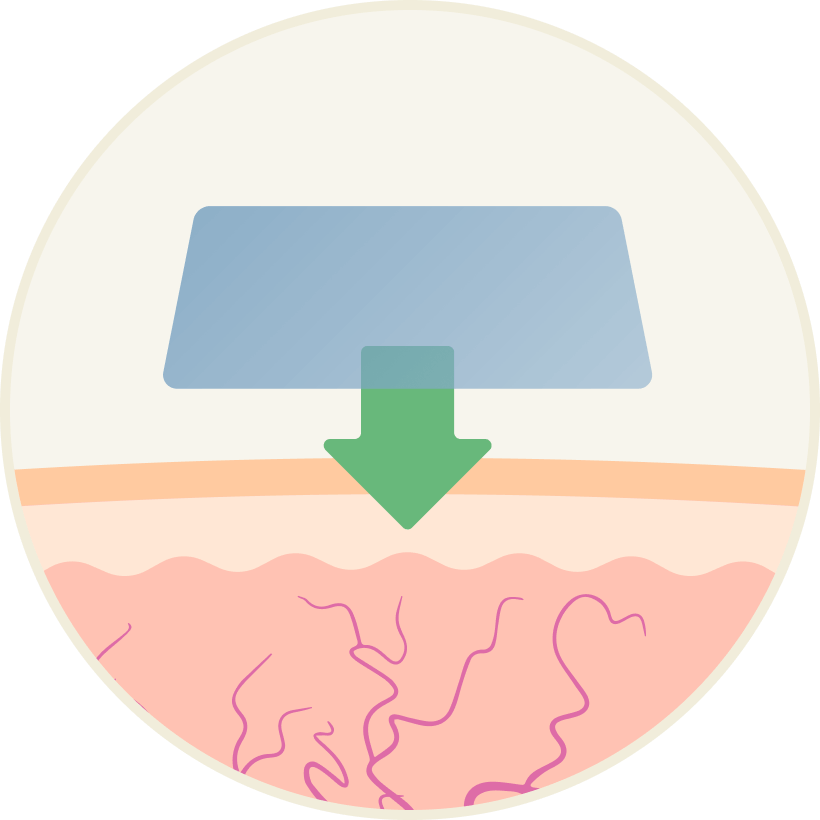
Klaria’s Alginate Film achieves transmucosal delivery. Active drug is transferred from the film, through the oral mucosa, directly into the blood. Importantly, the drug is not mixed with saliva, it is not swallowed and therefore it does not go into the stomach.
In this way, our technology is superior to all other oral transmucosal technologies such as films or sprays where there a small portion of the active drug is delivered transmucosally, but most of it ends up being swallowed and enters the stomach. Using such technologies, only drugs that are orally available can be used since most of the delivery is oral.
The proof? We focus on developing drugs that are not orally available and today have to be given as an injection. See more in our strategy section.
How it works
1) The alginate film is placed on the inside of the cheek and attaches immediately to the oral mucosa
2) Film stays in place for 5 minutes, then dissolves completely
3) The drug transfers directly into the bloodstream
Our Strategy

Our strategy is to develop new treatments where our technology creates maximum value. We do that by asking two questions:
Is treatment today available as an injection or a nasal spray?
Yes or No
If so, is this a significant problem for the patients, caregivers, doctors and/or payors?
Yes or No
If the answer to both of these questions is YES, then our technology solves important problems. We create a new product that is much better than the existing product and is easier and cheaper to make. Such an innovation would meet the definition of a Disruptive Innovation: a superior product made available at a lower price. This development would revolutionize the multi-billion dollar markets we are targeting.
Example: Adrenaline Alginate Film
Today, adrenaline is only available as an injection and is provided in a pre-filled auto-injector pen. The issues with the pens are many: they are difficult to give correctly, patients and caregivers are scared of injections, they are expensive, they are bulky and hard to carry around. Imagine if we can replace the auto-injector pen with a small and easy to carry film that is also easy to administer and in no way scary or painful. At the same time, the film is also much less expensive.
Our Adrenaline Alginate Film would meet the definition of a Disruptive Innovation: a superior product made available at a lower price. This development would revolutionize the multi-billion dollar market for adrenaline.
Pipeline

Sumatriptan
Klaria’s Sumatriptan Alginate Film is a novel treatment for Migraine. The film achieves transmucosal delivery of Sumatriptan, which has unique and valuable benefits as compared to the currently available formulations. This is especially true for the 80% of migraine sufferers who also suffer from nausea.
In 2018, Klaria received a grant of 21 MSEK in total from the innovation focused EU Horizon 2020 program. For more information, click here.
For more information about Sumatriptan Alginate Film and migraine, please click here.
Epinephrine/Adrenaline
The goals for Klaria's Adrenaline Alginate Film project are:
1. Replace EpiPen (aged incumbent technology with expensive auto-injector pen that is imprecise and bulky) with Epinephrine formulated into Klaria's Alginate Film
2. Disrupt the $4.3 billion/year product EpiPen with a superior product at a lower price for the benefit of all stakeholders
3. Claim substantially the entire market, making Epinephrine Alignate Film a massive commercial opportunity for Klaria
An important issue worth noting is that the adrenaline/epinephrine molecule suffers from very poor stability. Moreover, epinephrine is notoriously difficult to formulate and has until now only been available as an injection. Klaria’s successful formulation work to date is a testament to the power of our technology and the strong technical capabilities of our formulation scientists.
For more information about the Adrenaline Alginate Film project as well as allergic shock, please click here.
Sirolimus
Sirolimus has an immunosuppressant function and is used to prevent the rejection of organs in Solid Organ Transplantation (SOT) procedures. However, both the efficacy and safety of Sirolimus varies greatly between individual patients. This variability is due to how the treatment is metabolized. An Alginate Film that delivers Sirolimus directly into the blood stream via the oral mucosa would not be subject to this variability.
The result would be a treatment that would make life-saving Solid Organ Transplant procedures both less dangerous and more likely to succeed.

Cannabis Delivery Sciences
In October, 2019 Klaria announced the formation of daughter company Cannabis Delivery Sciences. The company is a separate entity focused on the commercialization of cannabis products formulated with Klaria’s Alginate Film Technology.
The mission of Cannabis Delivery Sciences is to create an entirely new, patent protected category of cannabis products. This new category of will combine the best qualities of the two currently available formulations of cannabis, edibles and smoking, without any of the drawbacks of either formulation.
To visit the website, click here.




